Nh4cl and water
Nh4cl And Water. Include the correct sign for the heat change. Improvements based on the literature were as follows. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40. Calculate the heat absorbed by the water or the solution in J and convert it to kJ.
 Ammonium Chloride When Dissolved In Water Leads To A Cooling Sensation The Dissolution Of Ammon Youtube From youtube.com
Ammonium Chloride When Dissolved In Water Leads To A Cooling Sensation The Dissolution Of Ammon Youtube From youtube.com
The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in the chemical industry. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40. Include the correct sign for the heat change. Ammonium salt and alkali reaction mechanism. In Part A the moles of NH4Cl are calculated from the mass of solid NH4Cl. Identify X and Y giving the chemical equation of the reactions involved.
Fill in the missing data in the following table.
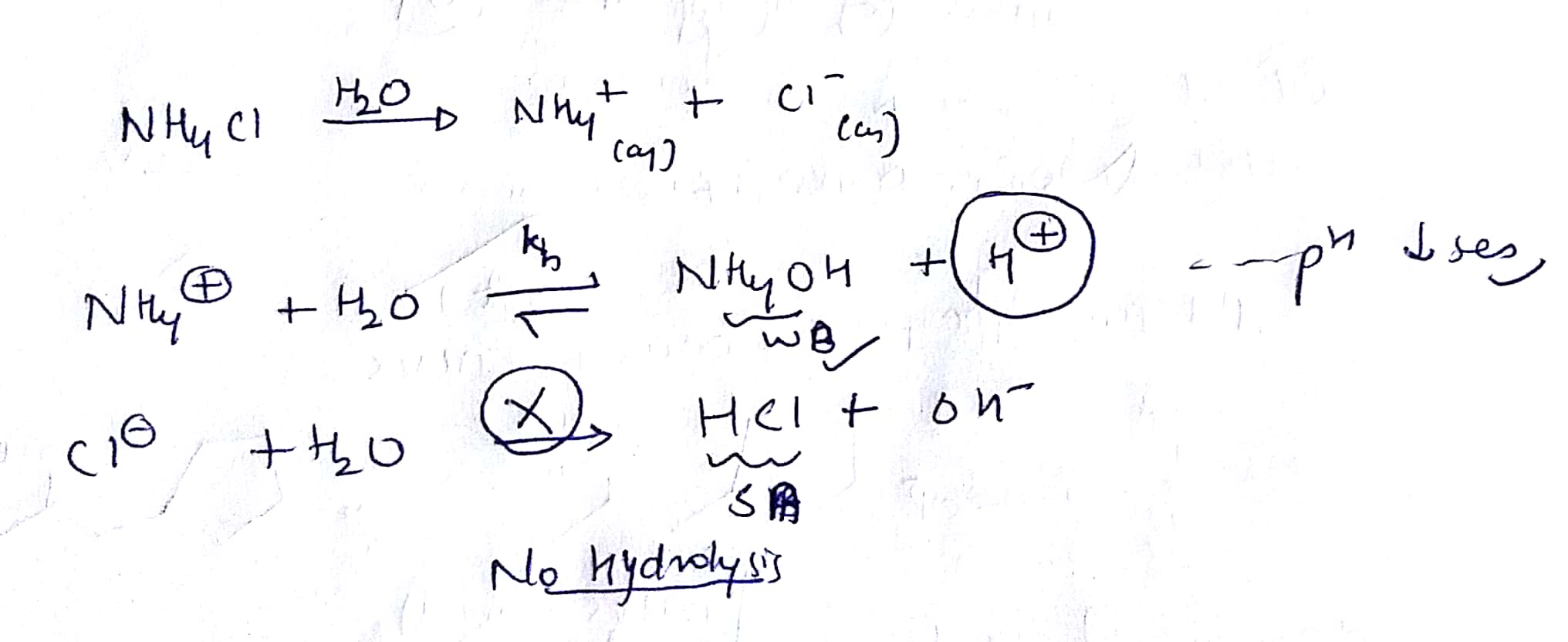
Aqueous ammonium chloride has weak acidic charactristics. Therefore electrons of oxygen atom in the hydroxyl ion attacks that hydrogen. Calculate the heat released by the reaction in terms of kJmol NH4Cl. Identify X and Y giving the chemical equation of the reactions involved. Hydrohen atoms of ammonium ion have a small positive charge. Milli-Q ultrapure water 18 MΩ cm 1 was used throughout the entire experimental project.
 Source: youtube.com
Source: youtube.com
The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in the chemical industry. Fill in the missing data in the following table. Identify X and Y giving the chemical equation of the reactions involved. In Part B the moles of NH4Cl are calculated from the volume and. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40.
 Source: youtube.com
Source: youtube.com
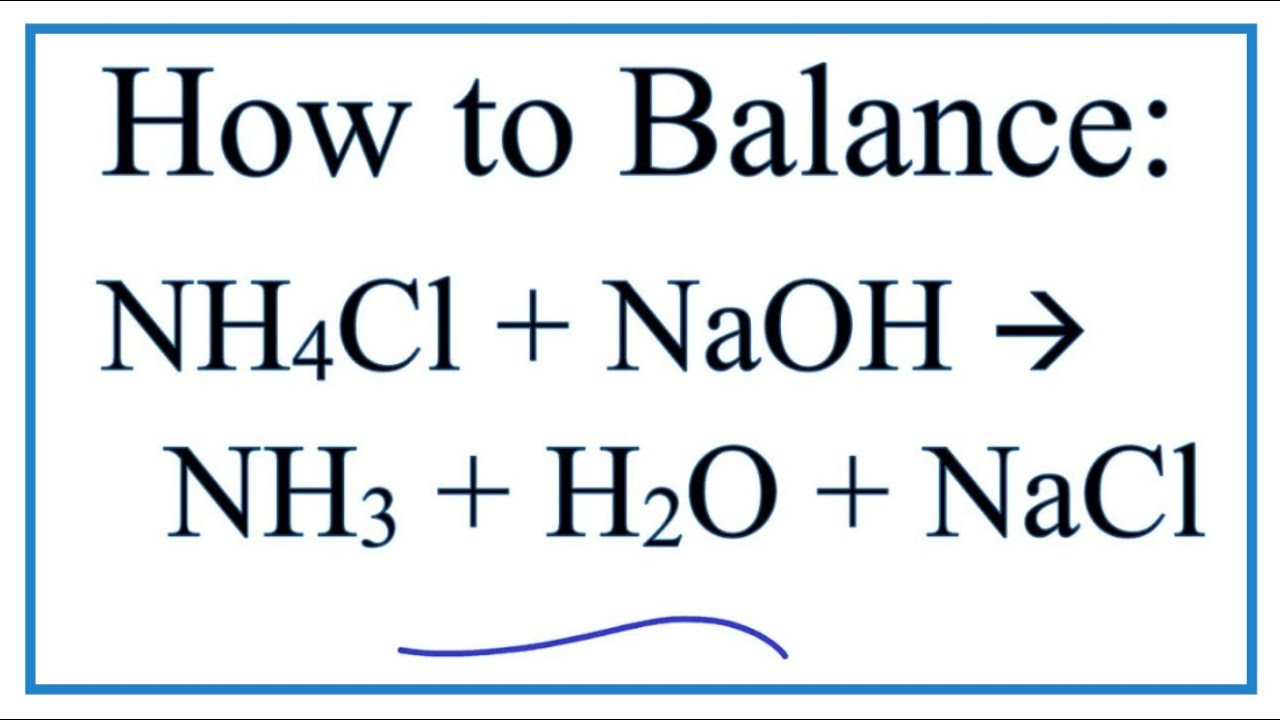
Include the correct sign for the heat change. Calculate the heat released by the reaction in terms of kJmol NH4Cl. Ammonia sodium chloride NaCl and water are given as products when ammonium chloride is heated with aqueous sodium hydroxide NaOH aq. Identify X and Y giving the chemical equation of the reactions involved. Fill in the missing data in the following table.
 Source: youtube.com
Source: youtube.com
First 5947 mg 3 mmol of 110-phenanthroline C 12 H 8 N 2 H 2 O was dissolved in 200 mL of hot. Improvements based on the literature were as follows. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40. Milli-Q ultrapure water 18 MΩ cm 1 was used throughout the entire experimental project. Fill in the missing data in the following table.
 Source: youtube.com
Source: youtube.com
First 5947 mg 3 mmol of 110-phenanthroline C 12 H 8 N 2 H 2 O was dissolved in 200 mL of hot. Ammonium salt and alkali reaction mechanism. Identify X and Y giving the chemical equation of the reactions involved. Milli-Q ultrapure water 18 MΩ cm 1 was used throughout the entire experimental project. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40.
 Source: youtube.com
Source: youtube.com
Calculate the heat absorbed by the water or the solution in J and convert it to kJ. First 5947 mg 3 mmol of 110-phenanthroline C 12 H 8 N 2 H 2 O was dissolved in 200 mL of hot. In Part B the moles of NH4Cl are calculated from the volume and. Calculate the heat released by the reaction in terms of kJmol NH4Cl. Ammonia sodium chloride NaCl and water are given as products when ammonium chloride is heated with aqueous sodium hydroxide NaOH aq.
 Source: chegg.com
Source: chegg.com
Ammonium salt and alkali reaction mechanism. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in the chemical industry. Hydrohen atoms of ammonium ion have a small positive charge. Calculate the heat released by the reaction in terms of kJmol NH4Cl. In Part B the moles of NH4Cl are calculated from the volume and.
 Source: numerade.com
Source: numerade.com
First 5947 mg 3 mmol of 110-phenanthroline C 12 H 8 N 2 H 2 O was dissolved in 200 mL of hot. Hydrohen atoms of ammonium ion have a small positive charge. In Part A the moles of NH4Cl are calculated from the mass of solid NH4Cl. In Part B the moles of NH4Cl are calculated from the volume and. Fill in the missing data in the following table.
 Source: embibe.com
Source: embibe.com
Therefore electrons of oxygen atom in the hydroxyl ion attacks that hydrogen. In Part A the moles of NH4Cl are calculated from the mass of solid NH4Cl. Aqueous ammonium chloride has weak acidic charactristics. Fill in the missing data in the following table. Hydrohen atoms of ammonium ion have a small positive charge.
 Source: youtube.com
Source: youtube.com
Fill in the missing data in the following table. The gas X reacts with lime water to give a compound Y which is used as a bleaching agent in the chemical industry. In Part A the moles of NH4Cl are calculated from the mass of solid NH4Cl. Fill in the missing data in the following table. Aqueous ammonium chloride has weak acidic charactristics.
 Source: chegg.com
Source: chegg.com
Fill in the missing data in the following table. Ammonium salt and alkali reaction mechanism. X Chlorine Cl 2 Y Bleaching powder CaOCl2 CaOH2 s Cl2 g CaOCl2 s H2O Calcium oxychloride bleaching powder 40. In Part A the moles of NH4Cl are calculated from the mass of solid NH4Cl. Milli-Q ultrapure water 18 MΩ cm 1 was used throughout the entire experimental project.
If you find this site serviceableness, please support us by sharing this posts to your favorite social media accounts like Facebook, Instagram and so on or you can also save this blog page with the title nh4cl and water by using Ctrl + D for devices a laptop with a Windows operating system or Command + D for laptops with an Apple operating system. If you use a smartphone, you can also use the drawer menu of the browser you are using. Whether it’s a Windows, Mac, iOS or Android operating system, you will still be able to bookmark this website.






